Anemia definition
Anemia definition
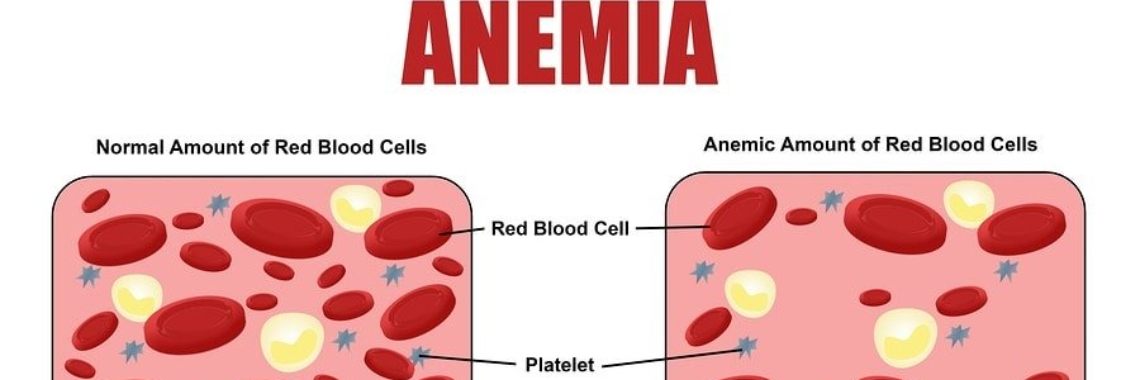
Iron deficiency anemia arises when the balance of iron intake, iron stores, and the body's loss of iron are insufficient to fully support production of erythrocytes. Iron deficiency anemia rarely causes death, but the impact on human health is significant. In the developed world, this disease is easily identified and treated, but frequently overlooked by physicians. In contrast, it is a health problem that affects major portions of the population in underdeveloped countries.
Overall, the prevention and successful treatment for iron deficiency anemia remain woefully insufficient worldwide, especially among underprivileged women and children. Here, clinical and laboratory features of the disease are discussed, and then focus is placed on relevant economic, environmental, infectious, and genetic factors that converge among global populations.
Nutritional anemias: iron, vitamins A and B12, folate, and riboflavin
Nutritional anemias result when concentrations of hematopoietic nutrients—those involved in RBC production or maintenance—are insufficient to meet those demands.13 Causes of nutrient deficiency include inadequate dietary intake, increased nutrient losses (e.g., blood loss from parasites, hemorrhage associated with childbirth, or heavy menstrual losses), impaired absorption (e.g., lack of intrinsic factor to aid vitamin B12 absorption, high intake of phytate, or
Helicobacter pylori infection that impair iron absorption), or altered nutrient metabolism (e.g., VA or riboflavin deficiency affecting mobilization of iron stores). While nutrient supplementation is a common preventive and treatment strategy for nutritional anemias—for example, iron supplementation for the prevention of IDA—the bioavailability and thus absorption from different nutrient supplement preparations can vary, potentially limiting their impact.53ID is considered the most common nutritional deficiency leading to anemia, though other nutritional deficiencies can also cause anemia, including deficiencies of vitamins A, B12, B6, C, D, and E, folate, riboflavin, copper, and zinc.54 Several of these nutrients—vitamins A, B6, and B12, folic acid, and riboflavin—are needed for the normal production of RBCs; other nutrients, such as vitamins C and E, may protect RBCs through their antioxidant function. Trace elements, such as copper and zinc, are found in the structures of enzymes that act on iron metabolism (e.g., copper and ceruloplasmin).56 Copper may also contribute to anemia development through reductions in erythropoietin (EPO) and antioxidant enzymes that require copper,